
(In fact, it is ionic.) In contrast, the compound NO 2 contains two elements that are both nonmetals (nitrogen, from group 5A, and oxygen, from group 6A). Therefore, it is most likely an ionic compound. For example, CaBr 2 contains a metallic element (calcium, a group 2A metal) and a nonmetallic element (bromine, a group 7A nonmetal). First, compounds between metal and nonmetal elements are usually ionic. There are two ways to recognize ionic compounds. This is to show that the subscript applies to the entire polyatomic ion. If more than one of a particular polyatomic ion is needed to balance the charge, the entire formula for the polyatomic ion must be enclosed in parentheses, and the numerical subscript is placed outside the parentheses. The rule for constructing formulas for ionic compounds containing polyatomic ions is the same as for formulas containing monatomic (single-atom) ions: the positive and negative charges must balance. The formula Mg 2Cl 4 has balanced charges with the ions in a 1:2 ratio, but it is not the lowest whole number ratio. Rather, it exists as two individual chloride ions.) By convention, the lowest whole number ratio is used in the formulas of ionic compounds. Chlorine does not exist as a diatomic element in this compound. (Do not read the “Cl 2” part of the formula as a molecule of the diatomic elemental chlorine. This chemical formula says that there are one magnesium ion and two chloride ions in this formula. We could write the chemical formula for this ionic compound as MgClCl, but the convention is to use a numerical subscript when there is more than one ion of a given type-MgCl 2. Now the positive and negative charges are balanced. The easiest way to balance these charges is to assume the presence of two chloride ions for each magnesium ion: A magnesium ion has a 2+ charge, while a chlorine ion has a 1− charge:Ĭombining one ion of each does not completely balance the positive and negative charges. Now consider the ionic compound formed by magnesium and chlorine. Therefore, the proper formula for this ionic compound is MgO. Although both of these ions have higher charges than the ions in lithium bromide, they still balance each other in a one-to-one ratio. When an ionic compound is formed from magnesium and oxygen, the magnesium ion has a 2+ charge, and the oxygen atom has a 2− charge. Only one ion of each is needed to balance these charges. If we look at the ionic compound consisting of lithium ions and bromide ions, we see that the lithium ion has a 1+ charge and the bromide ion has a 1− charge. Each ion is surrounded by ions of opposite charge.īy convention, assume that there is only one atom if a subscript is not present. Although it is convenient to think that NaCl crystals are composed of individual NaCl units, Figure 3.6 “A Sodium Chloride Crystal” shows that no single ion is exclusively associated with any other single ion. 
A macroscopic sample is composed of myriads of NaCl pairs each pair called a formula unit. Thus, NaCl is the chemical formula for sodium chloride, which is a concise way of describing the relative number of different ions in the compound. In the case of sodium chloride, the ratio of sodium ions to chloride ions, expressed in lowest whole numbers, is 1:1, so we use NaCl (one Na symbol and one Cl symbol) to represent the compound. However, we can use the ratio of sodium ions to chloride ions, expressed in the lowest possible whole numbers, as a way of describing the compound. As you can see, there are no individual NaCl “particles” in the array instead, there is a continuous lattice of alternating sodium and chloride ions. Ionic compounds exist as alternating positive and negative ions in regular, three-dimensional arrays called crystals ( Figure 3.6 “A Sodium Chloride Crystal”). To better understand what a chemical formula means, we must consider how an ionic compound is constructed from its ions. A chemical formulais a concise list of the elements in a compound and the ratios of these elements. We have already encountered some chemical formulas for simple ionic compounds. 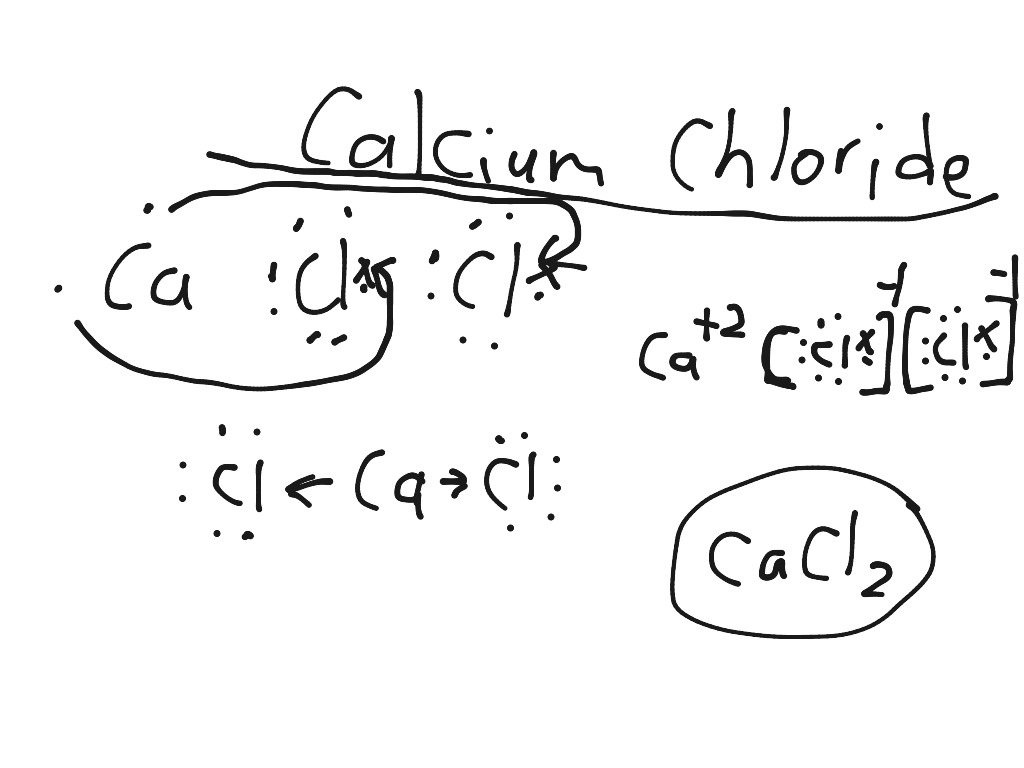
Recognize polyatomic ions in chemical formulas.Write the chemical formula for a simple ionic compound.
0 Comments
Leave a Reply. |
Details
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
